|
Protein binding is based upon hydrophobic interactions, as well as charge interactions between the membrane and protein. In order to make the proteins accessible to antibody detection, they are transfered by electroblotting from the gel onto a nitrocellulose membrane. Technical Note: Before beginning the transfer step, prepare the transfer buffer (1X with 10% methanol) and pre-cool to 4☌ in a cold room. In our laboratory, we have obtained good and reproducible results for various biochemical applications using this western-blotting method. The procedure described in this video article utilizes the Bis-Tris discontinuous buffer system with 4-12% Bis-Tris gradient gels and MES running buffer, as an illustration of how to perform a western-blot using the Invitrogen NuPAGE electrophoresis system. This system presents several advantages over the traditional Laemmli technique including: i) a longer shelf life of the pre-cast gels ranging from 8 months to 1 year ii) a broad separation range of molecular weights from 1 to 400 kDa depending of the type of gel used and iii) greater versatility (range of acrylamide percentage, the type of gel, and the ionic composition of the running buffer). It is an innovative neutral pH, discontinuous SDS-PAGE, pre-cast mini-gel system. In our laboratory, we have chosen to use the commercially available NuPAGE electrophoresis system from Invitrogen. Since its first description, the western-blotting technique has undergone several improvements, including pre-cast gels and user-friendly equipment. After transfer to a membrane, the target protein is probed with a specific primary antibody and detected by chemiluminescence. Protein separation according to molecular weight is achieved using denaturing SDS-PAGE. In this method, a target protein is detected with a specific primary antibody in a given sample of tissue homogenate or extract. If the specific antibody concentration of a given unpurified antibody preparation is unknown, we recommend using a concentration/purification kit and refer to the table below as a guideline.Western Blotting (or immunoblotting) is a standard laboratory procedure allowing investigators to verify the expression of a protein, determine the relative amount of the protein present in different samples, and analyze the results of co-immunoprecipitation experiments. Unpurified antibody preparations differ significantly in antibody concentration. Suggested dilutions for antibodies with no recommended dilution on the datasheet 
However, especially for polyclonal antibodies, when there is a change in the staining results between batches of the same antibody, we recommend performing another titration experiment. Many antibodies will have similar batch-to-batch consistency therefore, only one titration experiment is required in most cases. Each dilution should be tested on the same sample type to keep the same experimental conditions. For example, if a product datasheet suggests using a 1:200 dilution, it is recommended to make dilutions of 1:50, 1:100, 1:200, 1:400 and 1:500.Ī titration experiment is done by first selecting a fixed incubation time and then a series of experimental dilutions of the antibody. The optimal antibody concentration, which gives the best staining with minimum background, must be determined experimentally for each assay and is usually determined using a series of dilutions in a titration experiment. Optimizing the antibody dilution: titration experiments 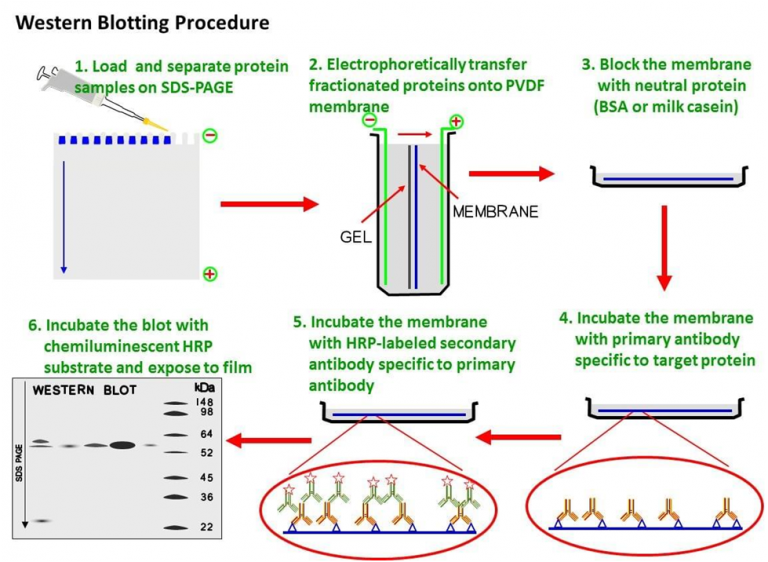
However, they may require some optimization in your specific experimental setup. 
Usually, antibodies have recommended dilutions for various applications included in the datasheet. As it is not usually possible to change the antigen concentration, the optimal working concentration of each antibody must be determined with dilutions for each application and set of experimental conditions. Varying the relative concentrations of an antibody and an antigen solution can also control the extent of antibody-antigen complex formation. The rate of binding between antibody and antigen – affinity constant – can be affected by temperature, pH, and buffer constituents. Why do you need to define antibody dilution for your experimental conditions?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
